Case Studies
Helping Clients Monitor Compliance Health
4 min read | Published

About
As the regulations in life science industries become more centerpiece, entire industries have arisen around the cause of helping companies comply with the new rules — and stay compliant.
MediSpend helps clients monitor compliance for pharmaceuticals, medical device, and biotechnology companies by collecting, tracking, and reporting all payments made to healthcare professionals.
Customer Since
2013
Use Case
Compliance in Life Sciences
Solution
MediSpend Insights Powered by GoodData
The Sunshine Act of 2013, requires pharmaceuticals, medical device, and biotechnology companies to collect, track, and report all payments made to healthcare professionals. “That means that every dollar these companies spend on healthcare professionals — from speaking engagement fees to research funds to the cost of the lunch they bring in for a meeting — has to be tracked and reported to the federal government, as well as to some state agencies,” explains Neeraj Singhal, Vice President Product Management for MediSpend.
Every time a company reimburses a healthcare professional, he or she generates what regulators call a “Transfer-of-Value”. The company must keep records of all such Transfers of Value and measure them against the thresholds based on their company’s policies and direction. Violation of policies and not conforming to compliance controls leads to serious fines sometimes to the tune of millions of dollars not to mention multi-year oversight of the company’s compliance processes and procedures by monitors that cost the company a lot of money. Life Sciences companies internationally including in the European Federation of Pharmaceutical Industries and Associations (EFPIA) face similar regulatory challenges.
“Keeping track of all these transactions manually would involve sifting through hundreds of Excel spreadsheets,” says Singhal. “All this would be prohibitively time consuming, not to mention carry a high risk of error. MediSpend was founded to help companies meet these requirements efficiently, and now we are providing embedded analytics into everyday workflow to help companies and compliance teams be more proactive in addressing problem areas.”
Seeking a Cure for Onboarding Ailments
MediSpend has partnered with GoodData since the product’s earliest iteration. “During the first few years,” explains Singhal, “we partnered with a third party for the ETL (extract, transform, and load) side and with GoodData for the analytics.”
Soon it became clear that having separate data management and dashboard technologies was sub-optimal and was not efficient in onboarding new customers. “We were having to do a lot of customization at the ETL level and had to create custom ETL and dashboards for every customer, even those who wanted a standard setup,” Singhal recalls. “The problem was not only the time delay, but also the cost of personnel, infrastructure, and other resources that the onboarding process required.”

The (Data) Doctor Is In
In 2016 Singhal and his team decided to use the GoodData platform for the entire end-to-end process. “By this point, we had been partnering with GoodData for years,” explains Singhal, “so we knew it was the right decision to use their entire offering from the product perspective and on the financiallevel.”
In the fall of 2016, the company released MediSpend Insights, with the actionable analytics embedded into the solution, alongside an updated version of the platform, MediSpend 4.0.
Best Features:
- Life Cycle Management (LCM)
- Visualizations
- Easily handles large volumes of data
Building Healthier Customer Relationships
MediSpend Insights is an optional upsell for users of the platform, and most new customers are opting for it. “Every customer we talk to is interested in analytics,” says Singhal. “They’re keen on not only seeing their data visually, but also being able to make decisions confidently based on suggestions they see in their customized dashboards; they want to manage based on facts. As a result, about 35 to 40 percent of our customer base is currently using the Insights platform.”
By consolidating its analytics process under GoodData, MediSpend has realized numerous benefits that not only improve its customers’ experience, but also benefit the company’s own bottom line.
Streamlined Onboarding
With the launch of MediSpend Insights, the company was able to leverage the benefits of GoodData’s Life Cycle Management (LCM) feature, which enables the configuration of multiple workspaces through master workspaces. “LCM has allowed us to streamline our onboarding process,” says Singhal. “It used to take days to get a new customer up and running, but with LCM, we can set up a standard dashboard in just a few hours.”
Results:
- 35-40% of customer base using MediSpend Insights
- Most new customers opt for the Insights upsell
- Reduced costs by eliminating separate ETL partner
- Cost reduction due to fewer resources needed for onboarding new customers
24-Hour Data Cycle
LCM has also accelerated the process of delivering daily data to customers. “We’re able to refresh the analytics every 24 hours or more frequently, as required,” Singhal explains. “It’s a fully automated, overnight process, so customers have access to up-to-date dashboards as soon as they come in every morning.”
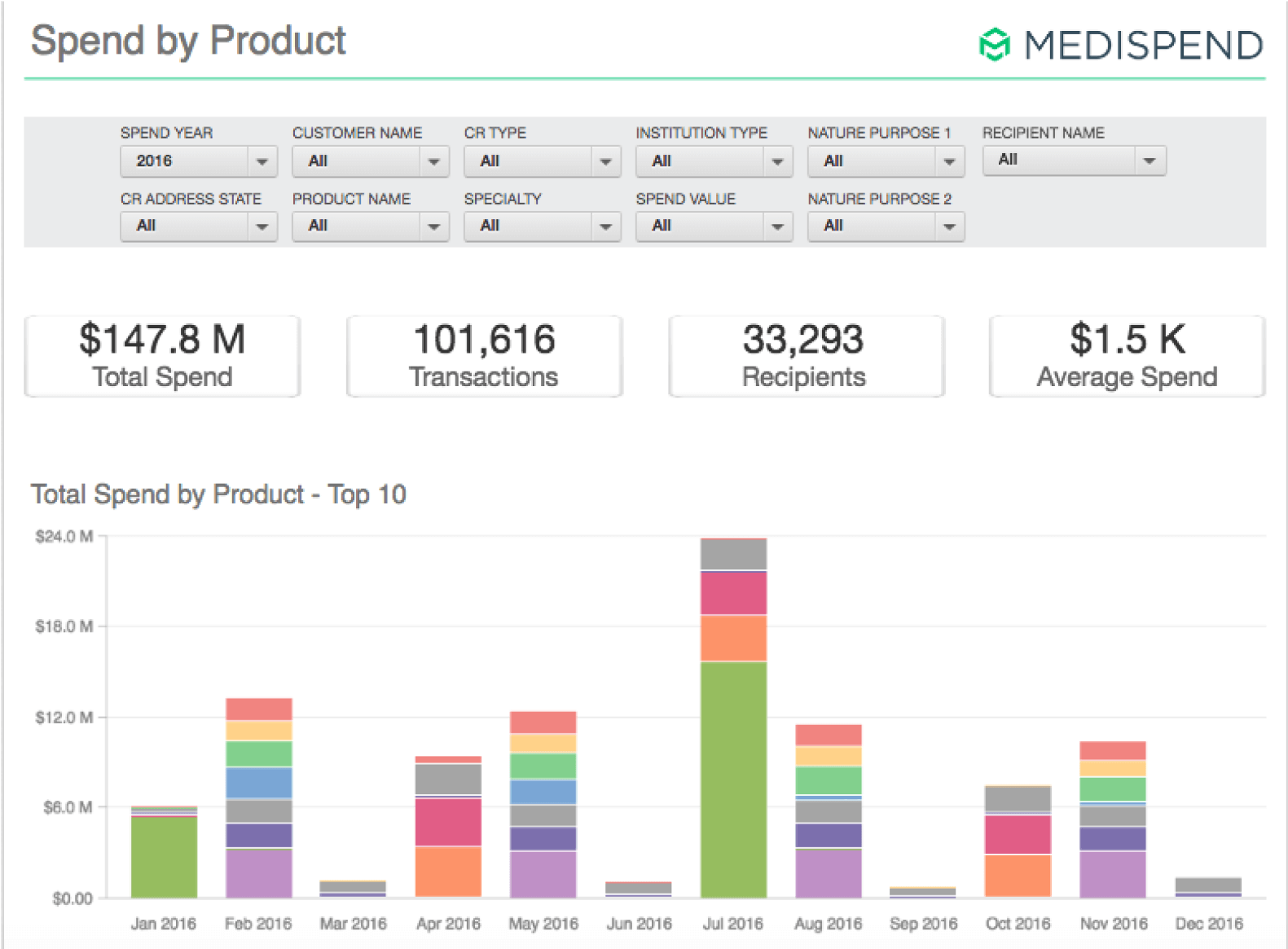
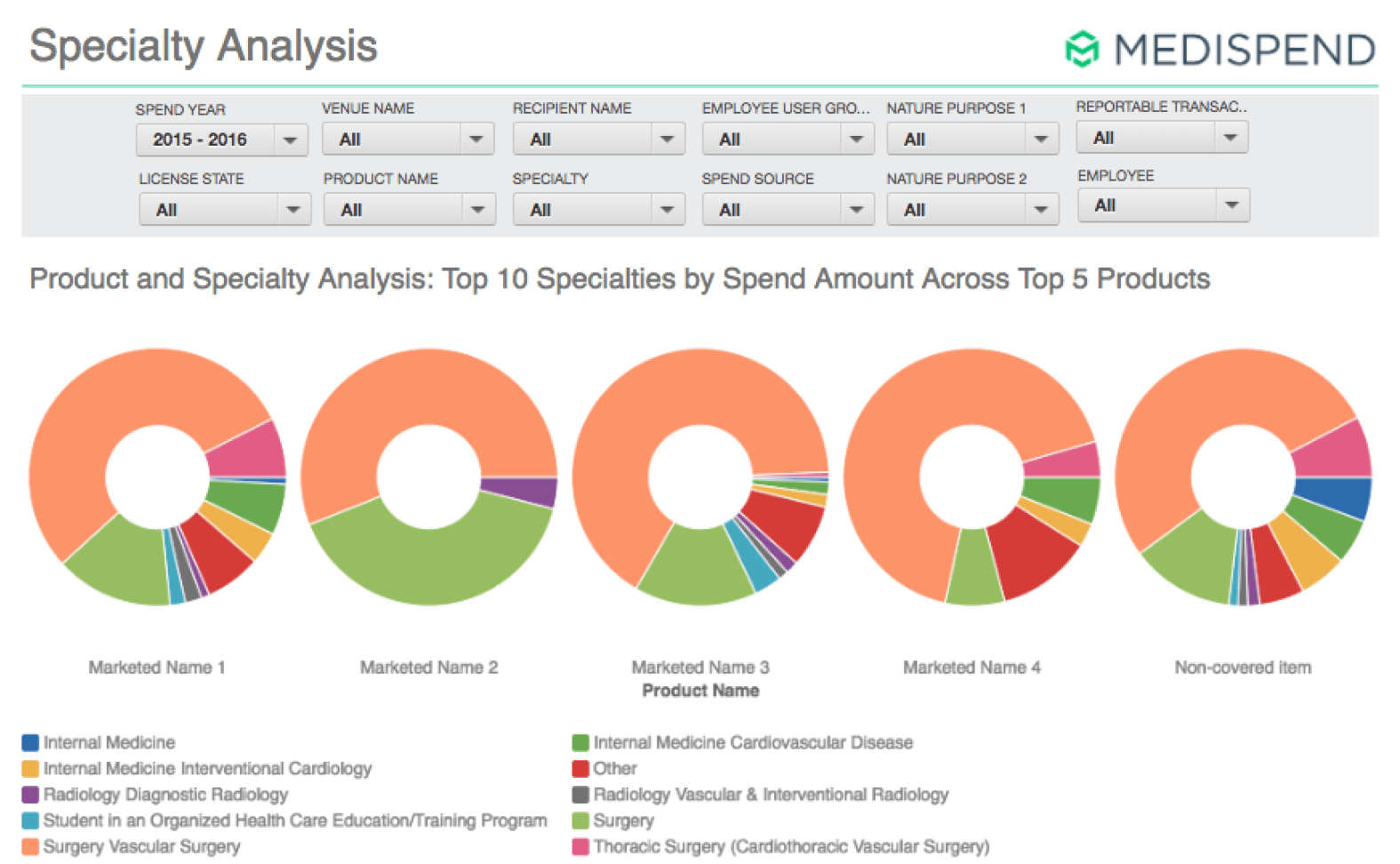
Reduced Expenses for Onboarding
Finally, the company has been able to reduce its own expenses around the customer onboarding process. Singhal notes that under the previous version of MediSpend, the need to manage multiple environments drove up their personnel and infrastructure costs. Now that the entire process is consolidated in the GoodData environment, fewer resources are needed and costs have thus gone down.
As the regulatory picture becomes increasingly complex for life science companies, MediSpend is seeing a growing array of opportunities. “As we continue to build up data,” Singhal notes, “we are looking at ways to increase the value we deliver to our customers. For example, one day we aim to offer recommendations and suggested actions to enable much more effective and efficient compliance monitoring. It’s another way of helping our customers stay one step ahead of the compliance game and address issues before they can turn into problems.”

Does GoodData look like the better fit?
Get a demo now and see for yourself. It’s commitment-free.







